| BIODIVERSITY AND SUSTAINABILITY | ||||||||
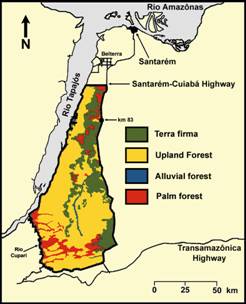
Neotropical Deforestation.--On a global scale, habitat loss associated with anthropogenic activities is increasing at an alarming rate. This is particularly true in tropical environs, which currently are less degraded than their temperate counterparts. However, logging pressure related to economic growth and global timber needs continue to threaten pristine Neotropical forests in remote regions of Amazonia. Over the last 30 years, low-intensity logging practices (as opposed to conventional selective harvesting, timber liquidation or clear cutting) that harvest 2-8 trees/hectare have become increasingly common in the Neotropics. Reduced-impact logging (RIL) is a modification of low-intensity logging that implements pre- and post-logging guidelines to protect seedlings, saplings, and small trees from injury, minimize soil damage, prevent unnecessary damage to non-target species, and protect critical ecosystem processes (e.g., hydrogeology and carbon sequestration). Felling and skidding techniques used in conventional selective logging practices can kill 27 trees of ≥ 10 cm dbh for every harvested tree. In contrast, RIL minimizes damage to surrounding forest while extracting a reduced number (2-4 trees/hectare) and volume (5-19 m3/hectare) of commercially valuable trees. After logging, forest is allowed to recover for several years through natural regeneration before the next harvest. The cyclic nature and low-impact of these harvests on the structure and composition of the forest may minimize negative effects on biodiversity and provide a renewable resource of economic value. From a conservation perspective, it is essential to determine consequences of RIL practices on the biota. This is especially critical because such practices rapidly are becoming an industry standard. Current estimates from of deforestation predict that within 50 years expansion of agriculture associated with soy and cattle industries will reduce forest cover in the Amazon by 40%, with some watersheds (i.e., Tapajós and Xingu) subject to > 65% reduction in forest cover. |
||||||||
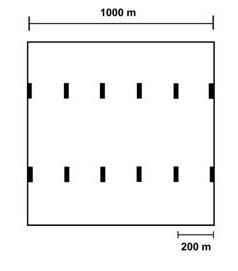
Bats as a Keystone Taxon.--In the Neotropics, bats provide critical ecosystem services associated with pollination and seed dispersal. Phyllostomid bats (New World fruit-eating bats) may represent a keystone taxon in Neotropical forests because they are the sole or primary agents of pollination and seed dispersal for many species of plants. Moreover, some phyllostomid species differentially disperse seeds of pioneer species of plants, which promotes secondary succession and regeneration of disturbed areas. In the Neotropics, seed dispersal by animals is integral to the establishment and persistence of a seed bank that is representative of the local flora. Consequently, understanding the effects of logging regimes on bat populations is critical to designing a forest management program that maintains a high degree of natural ecosystem function in lowland Amazonian rainforests. Bats respond to logging practices and associated habitat fragmentation in a species-specific manner; abundances of some species increase, others decrease, and yet others are unaffected. Few studies have evaluated the effects of low-intensity logging on bats and no studies previously evaluated effects of RIL practices on bat populations. Recent studies suggest that effects of RIL are minimal on non-volant mammals and birds, and that the detrimental effects of RIL are less severe than those of conventional logging techniques. To gain additional evidence concerning the effects of RIL on biodiversity, we designed a suite of analyses to evaluate effects of RIL on bats. More specifically, experiments were designed to evaluate effects of logging as well as effects of changes in forest physiognomy on populations, feeding guilds, and assemblages of bats. |
||||||||
|
||||||||
|
||||||||
|
||||||||
|
||||||||
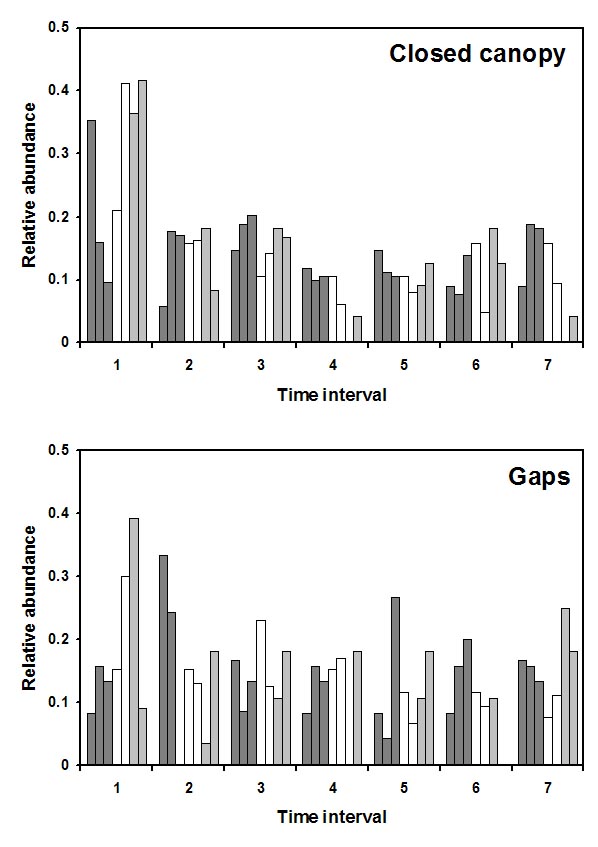
Results.--A total of 4,144 bats were recorded representing 55 species, 36 genera, 6 families, and 6 feeding guilds. Of the 15 species of bat that were sufficiently abundant for analysis, 10 species exhibited no response to RIL, 3 species (Artibeus concolor, Artibeus lituratus, and Glossophaga soricina) were more abundant in RIL forest, and two species (Carollia brevicauda and Carollia perspicillata) were more abundant in undisturbed forest. From a guild perspective, frugivores responded negatively to RIL; however, this was largely a result of the response Carollia perspicillata (the most abundant bat species in the TNF). Assemblage structure differed between RIL and undisturbed forest; assemblages in RIL forest had higher Shannon diversity and lower levels of dominance, but species richness, number of rare species, and evenness did not respond to RIL. Frugivores remained highly dominant in RIL forest. Indeed, RIL had little effect on guild abundance, guild composition, or guild structure. In general, responses of bat assemblage composition and structure to RIL were modest. These results suggest that RIL may have only small effects on ecosystem services provided by phyllostomid bats, at least in the short term.
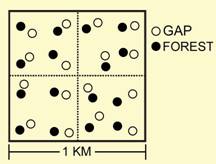
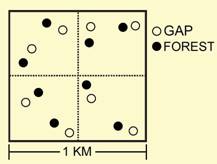
In the gap-matrix study, the effects of RIL and habitat type on abundance were evaluated separately for each of the 17 most abundant species of phyllostomid. Sixteen of 17 species responded to management (Glossophaga soricina, Carollia brevicauda, Rhinophylla pumilio, Artibeus gnomus, and Platyrrhinus helleri), habitat (Lonchophylla thomasi, Lophostoma silvicolum, Tonatia saurophila, Rhinophylla fischerae, Rhinophylla pumilio, Artibeus concolor, Artibeus planirostris, Artibeus obscurus, Platyrrhinus helleri, and Vampyressa bidens), or their interaction (Carollia perspicillata, Artibeus lituratus, and Uroderma bilobatum). Nine of the 10 species that responded to habitat in a consistent manner were more abundant in closed canopy sites than in gaps. In contrast, Rhinophylla fischerae was more abundant in gaps than in closed canopy sites. Regardless of habitat type, Rhinophylla pumilio was more abundant in undisturbed forest, whereas Glossophaga soricina, Carollia brevicauda, Artibeus gnomus and Platyrrhinus helleri were more abundant in RIL forest. Although common phyllostomids did not respond strongly to RIL from a quantitative perspective, in the gap-matrix study 16 phyllostomid species exhibited a negative qualitative response to RIL. Three common species were rare RIL forest, and 16 species that occur in undisturbed forest were not captured in RIL forest. However, some proportion of these observations likely are related to sampling effects (i.e., bias). |
||||||||
Implications of RIL.--Phyllostomids are the sole or primary seed dispersers or pollinators for many early successional plants; therefore, a significant reduction in the populations of these bats could dramatically affect forest regeneration and succession. In general, RIL did not reduce abundances of common frugivorous or nectarivorous phyllostomids, and sometimes increased their abundances. Clearly, increases in abundance do not represent conservation concern for the focal species, per se. However, the indirect consequences of such changes in abundance are unknown with respect to the suite of competitive, predator-prey, and mutualistic interactions that involve those species. Nonetheless, in general, common frugivorous and nectarivorous phyllostomids thrive in areas of limited human activity, such as in areas of RIL.
In contrast to the situation for common phyllostomids, many rare phyllostomids responded negatively to RIL. The fact that many species of rare phyllostomids were captured in control forest but not in RIL forest is a significant conservation concern. Moreover, several species exhibited negative responses (i.e., reduced abundances) to logging, even though they remained in RIL forest. These responses by rare species may represent short-term effects of RIL. High vagility of bats and close proximity of undisturbed forest which support these rare species may facilitate rescue effects that mitigate negative consequences of RIL. Reduced abundance of a species may signal a temporary reduction in carrying capacity, local migratory responses, or a more serious negative response to RIL that forebodes local extirpation. Of particular concern is loss of species that provide ecosystem services (e.g., frugivorous or nectarivorous bats) that could modify forest dynamics and successional trajectories. The ultimate consequence of poor survivorship of keystone taxa could be long-term degradation of forest that is incapable of providing renewable timber resources, thereby defeating the goal of RIL.
In addition to altering food availability, physical modification of the forest via removal of large trees during RIL may induce changes in bat populations by reducing the number and types of available roosts. About 72% of the bat species from the TNF use roosts associated with large trees (e.g., fallen logs, exfoliating bark, tree hollows, tree boles, dead standing trees, and ant/termite nests). Therefore, implementation of RIL which selects only large trees (i.e., trees with dbh > 45 cm) for harvest has the potential to decrease roost availability for many bat species. However, the average density of trees with dbh > 35 cm in TNF is 55 trees/ha; removal of < 4 trees per hectare represents a minor (7% or less) reduction in tree density. Such a reduction in roost availability may negatively affect abundances of some species, but likely is not sufficient to cause local extirpation. Nonetheless, careful examination of species-specific responses may reveal thresholds beyond which roosting habitat is too sparse for populations to persist. Because preferences for roosting habitat are unavailable for many Neotropical bat species, effects of long-term application of RIL on roost availability are unknown and worthy of future investigation. Although RIL may be a viable option to provide a renewable resource with minimal negative consequences for bats, the spatial arrangement of logging sites must be designed with care to minimize effects on regional biota. The 5000 hectare grid that defined our study area was associated with a largely undisturbed protected forest. This may reduce the impact of RIL on bats in TNF forest via rescue effects. Thus, the impact of RIL may be context dependent, and RIL may be sustainable from perspectives of biodiversity and ecosystem services only if large tracts of forest remain undisturbed in the vicinity of logged forest. |
||||||||
|
||||||||
Species of special concern.--Eight species that appear on the IUCN red list of threatened species were captured in TNF. Glyphonycteris sylvestris, Lophostoma carrikeri, Vampyrum spectrum, and Choeroniscus minor were rare; however, Rhinophylla fischerae, Artibeus concolor, Artibeus obscurus, and Vampyressa bidens were sufficiently abundant to quantitatively evaluate the effects of RIL and forest physiognomy on their abundances. Each of these species consistently responded to habitat type, with three species more abundant in closed canopy sites and one species (Rhinophylla fischerae) more abundant in gaps. None responded to RIL. That the Amazonian endemic Rhinophylla fischerae was more abundant in gaps (natural and man-made) suggests that gaps provide critical habitat for this restricted-range species. Nonetheless, because logging activity increases the proportion of the forest that is gap habitat and because most of the threatened species avoided gaps, dramatic increases in gap prevalence may adversely affect these populations and make them more prone to local extinction. Future directions.--As tropical forests suffer greater exposure to and use by humans, they increasingly will become smaller, fragmented, and exhibit a spatial configuration in which disturbance and successional recovery are dominant features of the landscape. As such, conservation strategies must optimally configure land uses to sustain natural populations and maintain the composition of local communities and regional assemblages, while accommodating the needs of society. In an attempt to create a sustainable and renewable resource, while minimizing impacts on biodiversity, conservation biologists and government agencies have endorsed harvest techniques (e.g., RIL) that minimize damages to surrounding forest that are associated with logging. A moratorium on logging most likely will not be an option, especially in the developing world. As such, it is imperative that conservation biologists identify viable options for sustainable use of forest that cause minimal impact on biodiversity. Otherwise, policy makers will make decisions based on purely economic concerns, without a balanced view tempered by conservation science. In short, forests will be cut to meet societal demands, better that it be managed with scientific guidance. This study was conducted between two and four years post-harvest and consequently represents only short-term responses to RIL. Compared to other logging techniques, the short term impact of RIL on bat populations, assemblages, and guilds was small, suggesting that RIL is a viable and potentially integral option for sustainable forest management. However, extrapolation of short-term consequences to a long-term prospectus is tenuous, and caution must be exercised in interpreting results in the context of conservation, management, or policy. Studies over longer periods of time or studies that include forest that was subjected previously to logging and is in various stages of regeneration provide greater insight into long-term implications of modern logging protocols. The selective harvest of trees in the Brazilian Amazon has increased in recent times, and likely will continue to increase. Consequently, long-term effects should be quantified to ascertain long-term consequences to biodiversity, as well as to ecosystem functionality and services. Evaluation of the long-term effects of RIL on keystone species is a crucial issue for evaluating sustainable use and conservation goals that are part of the expanded system of Brazil National Forests. Indeed, the efficacy of RIL may be tested in the near future: a conservation initiative recently enacted in Pará, Brazil only allows the use of sustainable logging and agricultural practices in seven areas in northern Amazonia that cover 15 million hectares. Because logging must be deemed sustainable to be executed within these expanded areas of conservation, clarification of the impacts of logging practices such as RIL on biodiversity is vital to the success of this new initiative and others like it.
|
||||||||
Collaborators on Reduced-Impact Logging Research
|
||||||||
Related Manuscripts Henriques, L.M.P., J.M Wunderle, & M.R. Willig. 2003. Birds of the Tapajos national Forest, Brazilian Amazon: a preliminary assessment. Ornithological Neotropical 14:307–338. Wunderle, J.M., M.R. Willig, & L.M.P. Henriques. 2005. Avian distribution in treefall gaps and understorey of terra firme forest in the lowland Amazon. Ibis 147:109–129. Wunderle, J.M., L.M.P. Henriques, & M.R. Willig. 2006. Short-term responses of birds to forest gaps and closed canopy: an assessment of reduced-impact logging in a lowland Amazon forest. Biotropica 38:235–255. Castro-Arellano, I., S. J. Presley, L. N. Saldanha, M. R. Willig, and J. M. Wunderle. 2007. Effects of low-intensity logging on bat biodiversity in terra firme forest of lowland Amazonia. Biological Conservation 138:269-285. Presley, S. J., M. R. Willig, J. M. Wunderle, and L. N. Saldanha. 2008. Effects of reduced-impact logging and forest physiognomy on bat populations of lowland Amazonian forest. Journal of Applied Ecology 45:14-25. Presley, S. J., M. R. Willig, L. N. Saldanha, J. M. Wunderle, and I.Castro-Arellano. 2009. Reduced-impact logging has little effect on temporal activity of frugivorous bats (Chiroptera) in lowland Amazonia.. Biotropica 41:369-378. Castro-Arellano I., Presley, S. J., M. R. Willig, J. M. Wunderle, and L. N. Saldanha. 2009. Reduced-impact logging and temporal activity of understorey bats in lowland Amazonia. Biological Conservation 142:2131-2139. |
||||||||
Please contact steven.presley@uconn.edu or michael.willig@uconn.edu if you have questions, comments, or corrections. Unless otherwise indicated, figures, tables, and images that appear on this page may not be used, reproduced, copied, or posted elsewhere, for any reasons, without permission. |