Difference between revisions of "Tobias Landberg"
| (3 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
| − | <span style="font-size: large"> | + | <span style="font-size: large">PhD</span><br> |
| − | + | '''E-mail:''' tlandberg@murraystate.edu <br> | |
| − | + | Starting in the fall of 2013, I'll be an assistant professor at [http://www.arcadia.edu/academic/biology/ Arcadia University] | |
| − | '''E-mail:''' | + | |
| − | ' | + | |
| − | + | ||
| − | + | ||
[[Image:Barbouribaby.jpg |center]]<br> | [[Image:Barbouribaby.jpg |center]]<br> | ||
== '''About''' == | == '''About''' == | ||
| − | + | This page is from my PhD in the Ecology & Evolutionary Biology department at the University of Connecticut, Storrs. I did my first post-doc with [http://people.bu.edu/kwarken/ Drs. Karen Warkentin] and [http://biology.vcu.edu/people/james-vonesh James Vonesh] studying red-eyed treefrogs at the Smithsonian Tropical Research Institute in Gamboa, Panama and a second post-doc in [http://campus.murraystate.edu/academic/faculty/hwhiteman/index.shtml Dr. Howard Whiteman's lab] working on environmentally induced polyphenisms in Ambystoma salamanders. <br> | |
| − | Co-advisors Drs. [http://hydrodictyon.eeb.uconn.edu/eebedia/index.php/Kurt_Schwenk Kurt Schwenk] & [http://hydrodictyon.eeb.uconn.edu/people/schlichting/ Carl Schlichting] headed my committee– which also included Drs. [http://hydrodictyon.eeb.uconn.edu/people/jockusch/jockuschlab/ Elizabeth Jockusch] and [http://hydrodictyon.eeb.uconn.edu/eebedia/index.php/Kentwood_Wells Kentwood Wells]. <br> | + | PhD Co-advisors Drs. [http://hydrodictyon.eeb.uconn.edu/eebedia/index.php/Kurt_Schwenk Kurt Schwenk] & [http://hydrodictyon.eeb.uconn.edu/people/schlichting/ Carl Schlichting] headed my committee– which also included Drs. [http://hydrodictyon.eeb.uconn.edu/people/jockusch/jockuschlab/ Elizabeth Jockusch] and [http://hydrodictyon.eeb.uconn.edu/eebedia/index.php/Kentwood_Wells Kentwood Wells]. <br> |
| − | + | ||
| − | |||
== '''Research philosophy''' == | == '''Research philosophy''' == | ||
| Line 129: | Line 125: | ||
<br> | <br> | ||
| − | |||
| − | |||
| − | |||
| − | |||
== '''Links''' == | == '''Links''' == | ||
The [http://hydrodictyon.eeb.uconn.edu/eebedia/index.php/Biology_Club Biology Club] is for anyone interested in undergraduate research in Biology at UConn. | The [http://hydrodictyon.eeb.uconn.edu/eebedia/index.php/Biology_Club Biology Club] is for anyone interested in undergraduate research in Biology at UConn. | ||
Revision as of 18:10, 27 April 2013
PhD
E-mail: tlandberg@murraystate.edu
Starting in the fall of 2013, I'll be an assistant professor at Arcadia University
Contents
About
This page is from my PhD in the Ecology & Evolutionary Biology department at the University of Connecticut, Storrs. I did my first post-doc with Drs. Karen Warkentin and James Vonesh studying red-eyed treefrogs at the Smithsonian Tropical Research Institute in Gamboa, Panama and a second post-doc in Dr. Howard Whiteman's lab working on environmentally induced polyphenisms in Ambystoma salamanders.
PhD Co-advisors Drs. Kurt Schwenk & Carl Schlichting headed my committee– which also included Drs. Elizabeth Jockusch and Kentwood Wells.
Research philosophy
In a nutshell, it's about unraveling the mobius strip of how organisms perform their behaviors, how sources of variation affect that performance, and how that performance affects evolution. Raising related species under a set of different environments can reveal how developmental responses to the environment evolve. Together with an understanding of the organisms' natural environments and the results of a variety of performance tests, functional variation can be used to interpret evolution.
Dissertation research
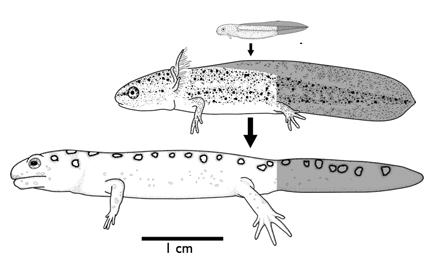
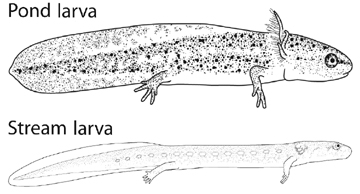
The broad goal of my dissertation is to see how maternal, developmental and environmental sources of variation contribute to species level diversity. The specifics include two types of experimental manipulations of environmental features. First, oxygen, because these salamander have an aquatic embryonic and larval phase to their life histories that spans a wide range of natural oxygen levels across streams and ponds. Second, maternal investment in yolk reserves, because female investment in egg size and number trade off. Investing in fewer offspring by the mother must gain an advantage for individual larvae. I'm looking at sister species that breed in ponds and streams (A. texanum & A. barbouri respectively).
Oxygen plasticity
Dissolved oxygen can act as a resource, because it is required for aerobic metabolism, and as information, because it can indicate poor water quality and drying pond conditions. Embryos raised in low oxygen were smaller than those raised in high oxygen indicating oxygen limits growth. The pond species, that lives in low oxygen habitats, had a much smaller response to oxygen than the stream species that generally experiences high oxygen. This suggests that the response to oxygen increases evolutionarily with the availability of oxygen. Later in development, pond larvae reversed the direction of their response so that larvae raised in low oxygen were larger than those raised in high oxygen. This growth response to low oxygen is correlated with early metamorphosis which suggests the response is adaptive when ponds dry up. The stream dweller, however, did not developmentally reverse the direction of plasticity. It continued having the largest body size in high oxygen throughout development. This pattern appears adaptive as well because the streams dry up quickly almost every year. Together these results suggest that the ancestral condition in pond species is oxygen limitation in embryos followed by adaptive plasticity in larvae. The stream species has evolved two adaptations of their response, a larger magnitude response and an reversal of the ancestral developmental reversal.
Maternal investment
Egg size is another feature that varies dramatically among amphibians living in different environments. Stream breeders typically have large eggs while pond breeders have relatively small eggs. This difference has important evolutionary consequences because females are resource limited and cannot both have large eggs and many of them. This trade-off and correlation with habitat strongly suggests that stream environments favor large eggs because of the high quality offspring they produce.
My approach to this question is experimental. By surgically removing small quantities of yolk from developing embryos, yolk reserves can be artificially manipulated. The strategy is to compare siblings who have had yolk removed to the sham operated group– those who were "poked" but no yolk actually removed. Tiny quantities of yolk can affect development from hatching all the way through metamorphosis.
Other salamander projects
During my tenure here at UConn, I've engaged in a number of other fruitful projects. They are just for fun and as such are all collaborative efforts.
Predator induced plasticity
One of the best-studied forms of adaptive plasticity in amphibians is their response to predators. Early hatching in response to egg predators is practically ubiquitous among amphibians, but what are the long term costs of leaving the egg early? How do predators that specialize on different life stages interact? Since metamorphosis is thought to be a particularly vulnerable life stage, is it responsive to the presence of predators? Has metamorphosis been selected to be as rapid as possible? Is it constrained physiologically by the demands of transformation? Together with Carl Schlichting, I have mentored two honors students who completed theses on some of these questions.
Leah Brown-Wilusz's honors thesis, was designed to address some of these questions in our local spotted salamander (Ambystoma maculatum). We raised embryos with and without egg predators (trichopteran larvae) and larval predators (marbled salamander larvae:Ambystoma opacum) to see if the early hatching response to one predator affected a future the response to larval predators. Presence of the egg predator caused early hatching regardless of the presence of the larval predator indicating that the immediate threat outweighs any future potential threat. Salamander embryos exposed to larval predators grew larger tail fins and survived longer with the predators but early hatching incurred a measurable cost. The effect of early hatching even extended to the adult life stage indicating that hatching early in response to an egg predator can make individuals smaller at metamorphosis. The early hatching response therefore looks to be maintained as a plastic strategy by costs in the larval period in terms of larval predator avoidance as well as potential fitness costs at metamorphosis.
Laurel Dwyer's honors thesis was designed to address several questions about the mysterious process of metamorphosis. We employed centrarchid sunfish (green sunfish: Lepomis cyanellus and bluegill sunfish: L. macrochirus) to see if spotted salamander larvae Ambystoma maculatum) can adaptively reduce their predation risk by reducing the amount of time they spend in the vulnerable process of metamorphosis. Behavioral trials indicated that predator exposed animals ate and weighed less explaining why they had lower survival despite only being exposed to chemical cues from the fish. Metamorphosis was approximately 25% shorter in duration for the salamanders exposed to predator cues compared to those not exposed. This suggests that salamanders assess their risk of predation and adaptively reduce the amount of time spent in the vulnerable metamorphic life stage.
Algae–salamander symbiosis
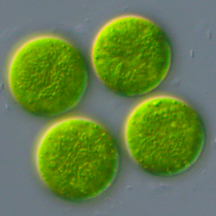
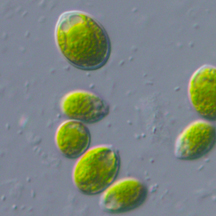
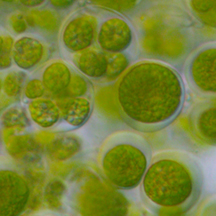
With undergraduate Shawn Binns, and professor Louise Lewis, I am investigating algae-salamander mutualisms. Ambystomatid salamander eggs deposited in vernal ponds often develop a green hue. This is not just a coating of pond scum, but rather an alga that lives inside the egg capsule. Poetically named, Oophila means egg-lover. Research has previously shown that the salamander benefits from increased oxygen levels produced by the algae while the algae gets nitrogenous waste products from the embryos. Everybody wins!
Who are these little green invaders? Do all the eggs have the same strain or species of algae. Does this vary from year to year? The algae inside of Louisiana spotted salamanders looks morphologically different from Connecticut algae. Is it plasticity or the same species? We also noticed that Kentucky algae growing on a different species (Ambystoma texanum) grows in the jelly matrix rather than inside the egg capsule. They look similar to the Connecticut algae but bigger. It's fun to think of salamander embryos as a source of food for plants.
Turtle research
Breathing & locomotion
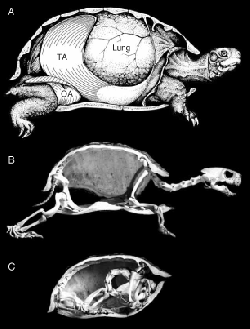
With Elizabeth Brainerd and Jeff Mailhot at the University of Massachusetts at Amherst, I studied turtle breathing and locomotion. Turtles can breathe using the limbs at rest... can they breathe during locomotion? Work done by Dr. Don Jackson and colleagues showed that nesting green sea turtles (Chelonia mydas) don't breathe at all during terrestrial locomotion. Box turtles and red eared sliders do however. Surprisingly, there is no relationship between limb movement and when breaths occur. So they can't be using the limbs for breathing during locomotion. Both species appear to use specialized abdominal muscles for breathing during locomotion, but there is one difference. There was no measurable effect of locomotion on breathing in the box turtle, Terrapene carolina- a species that has numerous adaptations for terrestriality. The red-eared slider, Trachemys scripta is much more typically semi-aquatic. When sliders pause between bouts of locomotion, they double the size of each breath indicating that locomotion interferes with breathing– by reducing the size of each breath. And the adult green sea turtle, of course, who has many highly derived features for aquatic locomotion, doesn't breathe during terrestrial locomotion. Three species–– three different life styles and three different patterns of interaction between breathing & locomotion. Makes you wonder...{{#ev:youtube|1fKBnE-_eC0}}
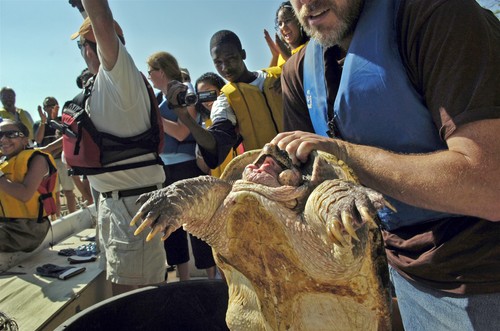
Snapping turtle ecology
In conjunction with many generous private volunteers, several local Connecticut organizations including Our Piece of the Pie, Riverfront Recapture, Connecticut Water Company as well as the National Geographic Society, I am working with high school students on a snapping turtle ecology. The goal is expose teenagers in Hartford, CT to nature, science and career options they might not otherwise see – and learn a thing or two about snappers.
Click the video below to see Shenipsit Lake, Connecticut from the shell of a 24 pound snapper!
{{#ev:youtube|EM2yj1_vBDE}}
Learn more about the snapping turtle research team
Publications
Landberg, T. and Schwenk, K. (in press). Book review: Steyermark, A.C. Finkler, M.S. & Brooks, R.J. (editors) 2008. Biology of the snapping turtle (Chelydra serpentina). – Baltimore, The Johns Hopkins University Press. Palarch’s Journal of Vertebrate Palaeontology
Landberg, T., Carlson, C. J., Abernathy, K., Luginbuhl, C. Gemme, P. and Mergins, C. (2010) Natural History Notes: CHELYDRA SERPENTINA SERPENTINA L. (Eastern Snapping Turtle). SURVIVAL AFTER INJURY. Herpetological Review 41(1):70-71 ![]()
Landberg T and Azizi, E. (2010) Ontogeny of escape swimming performance in the spotted salamander. Functional Ecology 24(3):576-586 ![]()
Landberg T., J. D. Mailhot and E. L. Brainerd (2009). Lung ventilation during treadmill locomotion in a semi-aquatic turtle, Trachemys scripta. Journal of Experimental Zoology 311A(8):551-562. ![]()
Azizi, E., T. Landberg and R. J. Wassersug (2007) Vertebral function during tadpole locomotion. Zoology 110:290-297. ![]()
Landberg, T., J. Mailhot and E.L. Brainerd (2003) Lung ventilation during treadmill locomotion in a terrestrial turtle, Terrapene carolina. Journal of Experimental Biology 206:3391-3404. ![]()
Azizi, E., and T. Landberg (2002) Effects of metamorphosis on the aquatic escape response of the two-lined salamander (Eurycea bislineata). Journal of Experimental Biology 205:841-849. ![]()
Links
The Biology Club is for anyone interested in undergraduate research in Biology at UConn.
My YouTube account has lots more videos
My Facebook account has lots more photos