Importance of considering multiple dimensions.-- The concept of biodiversity—the total variation of life—is multidimensional, including taxonomic, functional, phylogenetic, and genetic dimensions (Willig and Presley 2013). For the taxonomic dimension, taxonomic status is the only attribute of biodiversity. Other dimensions arise because salient differences between species are integral to the concept of biodiversity. More specifically, ecological, genetic, and evolutionary differences among species are inherent to the concept, and are embodied in functional, genetic, and phylogenetic dimensions. Each of these dimensions can be assessed with regard to only the presence or absence of species, or species can be weighted by some measure of their importance. Biodiversity research has predominately focused on the taxonomic dimension of biodiversity as measured by species richness as a proxy for all aspects of taxonomic biodiversity (e.g., evenness, dominance, dispersion, diversity) as well as for other dimensions of biodiversity (e.g., genetic, phylogenetic, functional). A consensus has emerged that taxonomic approaches do not adequately capture variation in other dimensions, with functional and phylogenetic dimensions often responding to environmental gradients differently than does the taxonomic dimension (Webb et al. 2002; Hooper et al. 2005; Safi et al. 2011; Cisneros et al. 2014).
The phylogenetic dimension of biodiversity reflects evolutionary differences among species based on times since divergence from a common ancestor (Faith 1992) and represents a comprehensive estimate of phylogenetically conserved ecological and phenotypic differences among species (Cavender-Bares et al. 2009). The functional dimension of biodiversity reflects variability in ecological attributes among species and provides a mechanistic link to ecosystem resistance, resilience and functioning (Petchey & Gaston 2006). The simultaneous assessment of variation in taxonomic, phylogenetic and functional dimensions of biodiversity along environmental gradients provides insights into the relative importance of ecological and evolutionary mechanisms that structure different components of assemblages.
Our interest in dimensions of biodiversity was inspired by participation in a distributed graduate seminar that included 10 institutions from the United States, China, Chile, Brazil, and Kenya. That seminar also spawned a blog entitled "BioDiverse Perspectives", which is run by graduates students from the seminar with the goal of fostering communication about biodiversity science among graduate students and throughout the scientific community.
| Fig. 1. Random selection is expected to produce positive non-linear relationships between phylogenetic or functional diversity and taxonomic diversity. |
Interdimensional relationships and structuring mechanisms.--Simultaneous consideration of multiple dimensions of biodiversity can provide evidence for the importance of particular ecological or evolutionary mechanisms in structuring communities. In general, random selection is expected to produce positive non-linear relationships between phylogenetic or functional diversity and taxonomic diversity (Kluge and Kessler 2011). Multiple ecological and evolutionary mechanisms can lead to functional or phylogenetic overdispersion (i.e. more functional or phylogenetic diversity than expected based on taxonomic diversity) or underdispersion (i.e. less functional or phylogenetic diversity than expected based on taxonomic diversity, Fig. 1). Communities that are dominated by some combination of competition among closely related species, partitioning of niches space, limiting similarity, or character displacement should have greater phylogenetic or functional diversity than expected based on taxonomic diversity. In contrast, communities that are dominated by some combination of environmental filtering (includes responses to the abiotic or biotic environment and interspecific interactions), competition among clades (Mayfield and Levine 2010), or trait convergence should have less phylogenetic or functional diversity than expected based on taxonomic diversity. Importantly, many of these processes always contribute to the structure of communities to some degree, but no single process may be sufficiently dominant to produce an effect distinct from expectations based on random selection. Consequently, random selection may be indistinguishable from a balance of processes associated with overdispersion and underdispersion.
| Fig. 2. Phylogenetic inertia gives rise to the a priori expectation that more closely related species will be functionally more similar, and more distantly related species will be functionally less similar. |
Phylogenetic inertia is the idea that no organism is re-designed from scratch, but always begins with characteristics inherited from ancestors (Shanahan 2011). Consequently, phylogenetic inertia gives rise to the a priori expectation that more closely related species will be more functionally similar, and more distantly related species will be less functionally similar (Safi et al. 2011). Competition within clades that leads to niche partitioning or limiting similarity, high environmental heterogeneity, or rapid trait evolution can result in greater functional diversity than expected based on phylogenetic diversity (Fig. 2). In contrast, environmental filtering (including responses to the abiotic or biotic environment and interspecific interactions), competition among clades, trait convergence, low environmental heterogeneity, or slow trait evolution can result in greater phylogenetic diversity than expected based on functional diversity. If opposing mechanisms are relatively balanced in effect, observations that are not distinguishable from phylogenetic inertia can result.
If a particular process or mechanism consistently dominates the assembly of an entire group of sites in a region, one would expect a strong relationship between dimensions (e.g. Petchey et al. 2007, Flynn et al. 2009, Kluge and Kessler 2011, Safi et al. 2011), with the form of the relationship providing evidence for a consistent lack of a dominant mechanism (e.g. random selection, phylogenetic inertia) or for mechanisms that results in overdispersion or underdispersion compared to expectations. In contrast, the lack of a significant relationship between dimensions may be evidence that the relative importance of mechanisms differs among sites, with the locations of particular sites in two-dimensional biodiversity space indicating the type of mechanism that dominates community assembly.
|
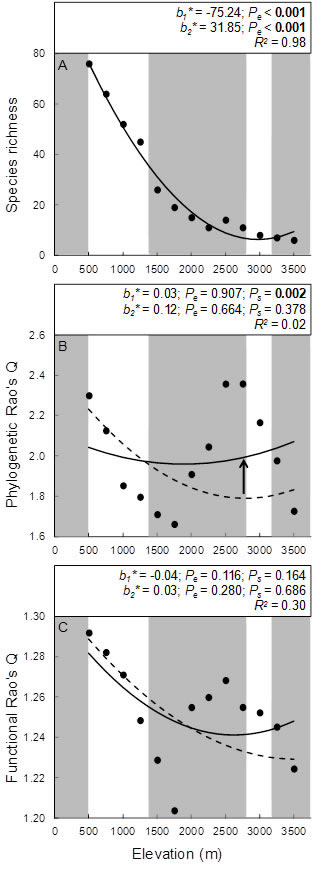
| Elevational variation in taxonomic, phylogenetic, and functional dimensions of biodiversity of bats at Manu (from Cisneros et al. 2014) |
Bats, rodents, and birds of Manu.--For each of 3 vertebrate orders (Chiroptera, Rodentia, Passeriformes), we assessed variation in taxonomic (species richness), phylogenetic, and functional dimensions of bat biodiversity along an extensive tropical elevational gradient in the Manu Biosphere Reserve. Manu is located along the eastern slopes of the Andes of southeastern Peru and covers overs 1,881,200 ha and elevations from 340 to 3625 m above sea level. Structurally distinct vegetation types replace one another along the elevational gradient, with lowland rainforest below 500 m of elevation, montane rainforest from 500 to 1400 m, cloud forest from 1400 to 2800 m, elfin forest from 2800 to 3200 m, and patches of elfin forest are intermixed with tall grasslands above 3200 m. Comprehensive data on the elevational distributions of 92 species of bat, 56 species of rodent, and 534 species of resident passerine from Manu were compiled from museum specimens, published literature, and recent surveys
Bats.--Bat species richness declined non-linearly with elevation, whereas phylogenetic dispersion and functional dispersion based on all functional components were not significantly associated with elevation. Moreover, considerable heterogeneity in the form of elevational relationships existed among functional components. Species richness was a poor surrogate for phylogenetic or functional dispersion. In addition, functional dispersion based on multiple components obscured patterns detected by particular components and hindered identification of mechanistic explanations for elevational variation in biodiversity. Variation in phylogenetic dispersion effectively captured the composite variation represented by all functional components, suggesting a phylogenetic signal in functional attributes. Mechanisms that give rise to variation in richness do not fully account for variation in phylogenetic or functional characteristics of assemblages. Greater than expected phylogenetic, diet, and foraging strategy dispersion at high elevations were associated with the loss of phylogenetically or functionally redundant species, suggesting that increasing interspecific competition with decreasing productivity resulted in competitive exclusion. In contrast, low dispersion of size attributes at high elevations suggests the importance of abiotic filtering that favors small-sized species that can more easily enter torpor (Cisneros et al. 2014).
Rodents.--Elevational gradients of each dimension of biodiversity were strong for rodents, with significant linear and non-linear components based on orthogonal polynomial regression. Empirical linear and non-linear regression components were significantly different than those expected based on species richness for generic, familial, and phylogenetic biodiversity, but not for functional biodiversity. In contrast to most studies, including bats from the same elevational gradient, analyses indicated that any one dimension would be a useful surrogate for the other dimensions. Given variation in species richness, assemblages from rainforests (300-1500 m) comprised more genera, families, body sizes, and evolutionary lineages than expected, whereas assemblages from cloud and elfin forests represented fewer genera, families, body sizes, and evolutionary lineages than expected. Warm temperature, vertical complexity of the vegetation, and high productivity likely facilitate niche differentiation in rainforests, whereas cricetid rodents are competitively superior to other clades in the less structurally complex, less productive, and colder, high elevation habitats (Dreiss et al. 2015).
|
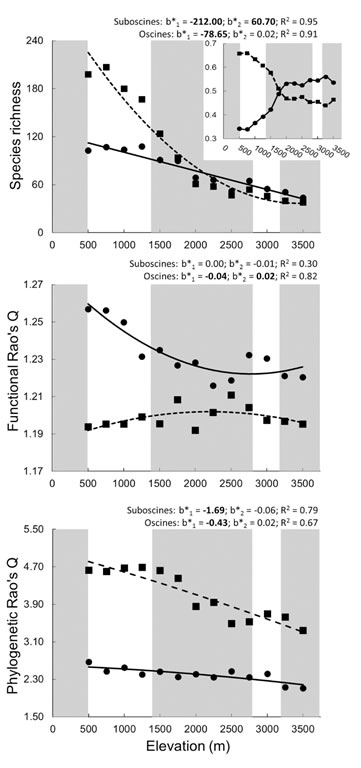
| Elevational variation in taxonomic, phylogenetic, and functional dimensions of biodiversity of passerines at Manu (from Burgio et al. submitted). Solid lines and dashed lines represent the best fit the quadratic relationship of oscine and suboscine biodiversity, respectively, with elevation. |
Passerines.--For passerines at Manu, elevational gradients were distinct for each dimension of biodiversity: taxonomic biodiversity decreased in a saturating fashion, phylogenetic biodiversity declined linearly, while functional diversity was stochastic (Burgio et al. submitted). Taxonomic and phylogenetic biodiversity were highly correlated, whereas functional biodiversity was not correlated with the other dimensions. The empirical elevational gradient of phylogentic biodiversity was significantly different from that expected based on species richness . In contrast, empirical elevational gradients of functional biodiversity were consistent with those expected based on species richness.
This study highlights the importance of historical biogeography and historical contingency for understanding extant patterns of biodiversity. Elevational gradients of biodiversity represented a combination of adaptations from radiations that occurred elsewhere (suboscines in Amazonian lowlands, oscines in colder climes of North America) and an in situ radiation (tanagers). Assemblages of volant vertebrates along the elevational gradient likely are molded by similar combinations of ecological mechanisms, most importantly physiological constraints associated with temperature, and a reduction in functional redundancy associated with decreasing resource abundance and diversity.
Phylogenetic Signals in Functional Traits.--In Prep
An Integrated Framework for Biodiversity.--One of the challenges we have encountered in this work is the identification of reliable metrics for phylogenetic or functional diversity that exhibit desirable behaviors and that accurately represent the concepts they are used to measure. In addition, comparisons with other studies have been confounded by use of metrics with undefined units, different units, or a lack of conceptual clarity concerning the properties measured by different metrics. We have been working with Sam Scheiner to further develop an integrated framework (Scheiner 2012) based on Hill numbers (Hill 1973) that uses the same conceptual approach to measure biodiversity for each dimension (Presley et al. 2014). This framework is promising for the development of diversity metrics that have desirable properties (e.g. the replication principle) and explicit units that facilitate ecologically meaningful comparisons among dimensions and among studies. In addition, this framework has two unique attributes that make it a powerful approach for considerations of biodiversity: (1) new metrics that measure different aspects of each dimension of biodiversity or new dimensions of biodiversity can be developed relatively easily, and (2) multiple aspects or dimensions of biodiversity can be integrated into a single measure.
|
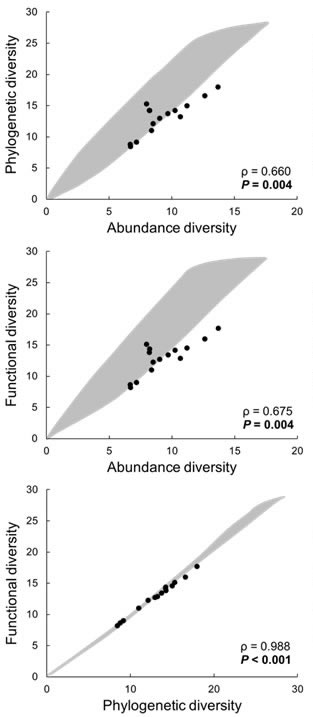
| Relationships between abundance, phylogenetic, and functional diversity for bats from the Caribbean lowlands. Shaded areas represent 95% confidence envelopes for pairwise considerations of dimensions (from Presley et al. submitted). |
To demonstrate the ease with which new metrics can be developed and incorporated into the framework, we introduced of an alternative functional diversity metric that measures functional dispersion (Presley et al. 2014). Using a comprehensive approach, one might wish to independently estimate diversity based on different niche axes (e.g., diet, foraging method, habitat use, masticatory mode, body size). These measures could then be combined into a single value of functional diversity.
Similarly, “importance” diversity could be separately calculated based on number of individuals, biomass, and frequency of occurrence (following the traditional use of importance values developed in the vegetation literature), and then integrated into a single metric that simultaneously accounts for each of these ways of being biologically “important”. Such separate and integrated measures may be useful in testing theories about the myriad ecological and evolutionary processes responsible for patterns of biodiversity.
Testing for mechanisms of phylogenetic and functional structure.--Using the integrated framework of biodiversity (Scheiner 2012) and the functional dispersion metric developed in Presley et al. (2014), we applied a Monte Carlo simulation to create 95% confidence envelopes for combinations of dimensions of biodiversity to detect deviations from expectation based on random sampling or phylogenetic inertia (Presley et al. submitted). To evaluate the effects of Neotropical forest loss and fragmentation from anthropogenic activities, we used data for Neotropical bats an extensively modified region (Caribbean lowlands of Costa Rica) and a relatively pristine area (Amazonian lowlands near Iquitos, Peru).
Most assemblages from Caribbean lowlands exhibited phylogenetic underdispersion, functional underdispersion, or functional deficit, with the remaining assemblages having structures consistent with null expectations. No assemblages exhibited phylogenetic overdispersion, functional overdispersion, or functional surplus. In Amazonian lowlands, all assemblages had phylogenetic and functional structure consistent with random selection, but most assemblages exhibited a functional deficit.
Extensive habitat loss in Caribbean lowlands created biotic filters (changes in habitat structure; changes in resource type, diversity, and abundance) that affect local species assembly, resulting in phylogenetic and functional underdispersion. In contrast, bat assemblages from relatively pristine lowland Amazonia exhibited structure consistent with random selection that likely arises from a balance of mechanisms that promote phylogenetic or functional overdispersion versus those that promote underdispersion.
Collaborators on Dimensions of Biodiversity Research
| Lab Members | Other Collaborators | ||
Related Manuscripts
Willig, M.R., and S.J. Presley. 2013. Latitudinal gradients of biodiversity. Pages 612-626 in Encyclopedia of Biodiversity, 2nd edition, Volume 4 (S. Levin, Ed.). Academic Press, Waltham, Massachusetts.
Cisneros, L.M., K.R. Burgio, L.M. Dreiss, B.T. Klingbeil, B.D. Patterson, S.J. Presley, and M.R. Willig. 2014. Multiple dimensions of bat biodiversity along an extensive tropical elevational gradient. Journal of Animal Ecology 83: 1124-1136. .
Dallas, T., and S.J. Presley. 2014. Relative importance of host environment, transmission potential, and host phylogeny to the structure of parasite metacommunities. Oikos 123: 866-874.
Presley, S.J., S.M. Scheiner, and M.R. Willig. Evaluation of an integrated framework for biodiversity with a new metric for functional dispersion. PLoS ONE 9(8): e105818. doi:10.1371/journal.pone.0105818.
Dreiss, L.M., K.R. Burgio, L.M. Cisneros, B.T. Klingbeil, B.D. Patterson, S.J. Presley, and M.R. Willig. 2015.Taxonomic, functional, and phylogenetic dimensions of rodent biodiversity along an extensive tropical elevational gradient. Ecography doi:10.1111/ecog.00971.
Cisneros, L.M., M.E. Fagan, and M.R. Willig. Effects of human-modified landscapes on taxonomic, functional and phylogenetic dimensions of bat biodiversity. Diversity and Distributions doi:10.1111/ddi.12277.
Presley, S.J., T. Dallas, B.T. Klingbeil, and M.R. Willig. 2015. Phylogenetic signals in host-parasite associations for Neotropical bats and Nearctic desert rodents. Biological Journal of the Linnean Society doi: 10.1111/bij.12601.
Presley, S.J., L.M. Cisneros, C.L. Higgins, B.T. Klingbeil, S.M. Scheiner, and M.R. Willig. Extensive habitat conversion at the regional scale creates environmental filters that mold functional and phylogenetic structure of Neotropical bat assemblages. Diversity and Distributions (submitted).
Burgio, K.R., L.M. Cisneros, K.E. Davis, L.M. Dreiss, B.T. Klingbeil, B.D. Patterson, S.J. Presley, and M.R. Willig. Dimensions of passerine biodiversity along a tropical elevational gradient: a nexus for historical biogeography and contemporary ecology. Ecography (Submitted).
References
Cavender-Bares, J., K. H. Kozak, P. V. A. Fine, and S. W. Kembel. 2009. The merging of community ecology and phylogenetic biology. Ecology Letters 12: 693–715.
Faith, D. P. 1992. Conservation evaluation and phylogenetic diversity. Biological Conservation 61: 1–10.
Flynn, D.F.G. M. Gogol-Prokurat, T. Nogeire, N. Molinari, B. Trautman Richers, B. B. Lin, N. Simpson, M. M. Mayfield, and F. DeClerck. 2009. Loss of functional diversity under land use intensification across multiple taxa. Ecology Letters 12: 22–33.
Hill, M. O. 1973. Diversity and evenness: a unifying notation and its consequences. Ecology 54: 427–432.
Hooper, D. U., F. S. Chapin, J. J. Ewel, A. Hector, P. Inchausti, S. Lavorel, J. H. Lawton, D. M. Lodge, M. Loreau, S. Naeem, B. Schmid, H. Setälä, A. J. Symstad, J. Vandermeer, and D. A. Wardle. 2005. Effects of biodiversity on ecosystem functioning: A consensus of current knowledge. Ecological Monographs 75: 3–35.
Kluge, J., and M. Kessler, M. 2011. Phylogenetic diversity, trait diversity and niches: species assembly of ferns along a tropical elevational gradient. Journal of Biogeography 38:394–405.
Mayfield, M. M., and J. M. Levine. 2010. Opposing effects of competitive exclusion on the phylogenetic structure of communities. Ecology Letters 13: 1085–1093.
Petchey, O. L., and K. J. Gaston. 2006. Functional diversity: back to basics and looking forward. Ecology Letters 9: 741–758.
Petchey, O. L., K. L. Evans, I. S. Fishburn, and K. J. Gaston. 2007. Low functional diversity and no redundancy in British avian assemblages. Journal of Animal Ecology 76: 977–985.
Safi, K., M. V. Cianciaruso, R. D. Loyola, D. Brito, K. Armour-Marshall, and J. A. Diniz-Filho. 2011. TPI 7469695 D15 Understanding global patterns of mammalian functional and phylogenetic diversity. Philosophical Transactions of the Royal Society B: Biological Sciences 366: 2536–2544.
Scheiner, S. M. 2012. A metric of biodiversity that integrates abundance, phylogeny, and function. Oikos 121: 1191–1202.
Shanahan, T. 2011. Phylogenetic inertia and Darwin’s higher law. Studies in History and Philosophy of Biological and Biomedical Sciences 42: 60–68.
Webb, C. O., D. D. Ackerly, M. A. McPeek, and M. J. Donoghue. 2002. Phylogenies and communit ecology. Annual Review of Ecology and Systematics 33: 475–505.