Snapping Turtle Research: Analyses and Conclusions
Below are the results of the 2008-2009 analysis of turtle footage collected by the Snapping turtle research team. All data was taken from three turtles - Jawless (39 lbs, no lower jaw), Snippy (24 lbs) and Lafayette (16 lbs). Lafayette and Jawless were both taken from Wethersfield Cove; Snippy was taken from Shenipsit Lake. Questions should be directed to Colin Carlson or Tobias Landberg.
Contents
Background
Locomotion: Extensive research has been done on the locomotion of other turtles, ranging in size from box turtles to green sea turtles. Limb cycle frequencies in aquatic systems have been reported as reaching up to 2.70 Hz (cycle/s) in Chelonia mydas, 1.7 Hz in Mauremys caspica, 1.0 Hz in Chrysemys scripta and 1.39 Hz in Kinosternon subrubrum in one particular study (Davenport et al., 1984)[1]. More recent studies of terrestrial turtle locomotion have suggested that adult Chrysemys picta may reach speeds up to 1.605 Hz voluntarily, and when forced, can even reach 2.557 Hz (Zani & Clausen, 1994) [2]. There appears to be something of a strong relationship between the typical size of a turtle species and its apparent rate of locomotion, something that will be tested in this research by evaluating where snapping turtles fit in this data set.
Quantified Behaviors
To describe the turtles' behavior, I've classified the turtles' activity into six major behaviors: breathing, horizontal swimming, vertical swimming (up and down), underwater walking, and pausing. Breathing involves surfacing and projecting the tip of the head out of the water to breathe; the turtle does not, however, extend its entire neck out of the water. In Jawless, this behavior is typically prefaced by a bout of horizontal locomotion, in which the turtle simply moves at a constant depth through the water; in contrast, Snippy's breathing generally followed a vertical dive to the bottom of the water, a bout of horizontal locomotion along the bottom (possibly with "underwater walking" interspersed with swimming) and a vertical dive up to the surface of the water. Jawless demonstrated underwater walking relatively rarely, and Snippy only demonstrated two major bouts; while the turtles probably did walk more than it appears on camera, the limbs often aren't visible while the turtle is on the bottom, preventing us from telling whether the turtle is actually walking; in such situations, we assumed that the turtle was in fact swimming.
In order to analyze these behaviors quantitatively, other related factors were brought into play: for instance, the amount of time the turtle spent during one "bout" of a particular behavior (which was often reduced to the amount of time visible on the camera, due to unclear or murky footage) was a useful measurement that helped determine each individual turtle's most common behavior. Another more quantifiable analysis was of limb cycle frequency, which was measured as (number of cycles)/(time), or in short, cycles/s. A cycle was defined as a movement of the front right limb, followed by a movement of the front left limb.
Breath-Dive Analysis
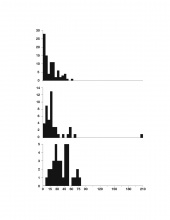
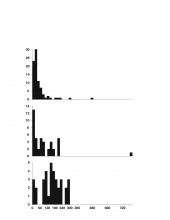
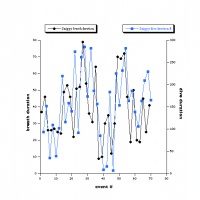
The results of this analysis, which are summarized in Fig. 1 and 2, were different for all three of the turtles, yet there are shared trends in their breathing and diving behavior, and these trends indicate a relationship between these two behaviors. Most importantly, there was a strong statistical relationship between breath and dive duration.
Research on factors influencing respiration in other turtles suggests that oxygen levels may be a more important factor than carbon dioxide buildup (for instance, Jackson 1985 on Chelonia mydas) [3]. Some species of turtle have also adapted to minimize response to either chemical cue, in particular, Caretta caretta (Lutz & Bentley, 1985) [4]. Based on the respiratory physiology of C. serpentina, some literature suggests that the partial pressure of carbon dioxide is the primary physiological/chemical factor influencing breathing frequency, and likely, breath duration (for example, West et al., 1989). [5]. However, there are an equal number of papers that suggest that oxygen levels are the limiting factor (see Bagatto & Henry, 1999) [6].
Thus, for this particular turtle, a likely hypothesis to explain this relationship between breath and dive duration based on West et al. 1989 would be that after a long dive, there is a large build-up of carbon dioxide that stimulates an impulse to breathe (I will refer to this as the "Anoxia Response Hypothesis", or ARH). An alternative hypothesis (implied by Bagatto & Henry, 1999) would be that breaths are "pre-emptive," and are intended to stock up on more oxygen for longer dives (the "Anoxia Preemption Hypothesis", APH). (Note that the null hypothesis, that there is no relationship between breathing and diving, has effectively been disproved.) Were the ARH true, one would expect there to be a stronger statistical relationship with dive duration for breaths following dives than breaths preceding dives; conversely, if breaths preceding dives were more strongly correlated with duration than breaths following dives, this would imply the APH to be true. However, the statistical relationship with dive duration is equally strong for breaths before and after dives. Thus, not only is there no general consensus on whether carbon dioxide or oxygen is more important in physiological cues for respiration, but the data set is also inconclusive.Another important hypothesis that could be created for this data pertains to the turtles' ecology. Note that the histograms reveal important information regarding the differences between the turtles. For both breath and dive duration, Jawless's distribution is extreme and somewhat exponential (this was a product of the numerous short, i.e. less than 1s to 3s, breaths at the very beginning of the footage, when Jawless's escape response pattern was most thoroughly demonstrated). On the other hand, Snippy's is almost perfectly symmetrical and unskewed, and is a relatively perfect bell curve. Lafayette falls somewhere inbetween - although not as exponential as Jawless's graph, Lafayette's chart is still centered further to the left than Snippy's, and is much more similar to Jawless's than to Snippy's. Although one would certainly expect some inter-turtle variation, why are the differences in the shapes of these histograms so pronounced?
One potential explanation of this difference is based on the turtles' respective habitats: if deeper water obliges vertical swimming, and vertical swimming takes more energy, then the turtles in deeper water have to breathe more to replenish oxygen levels. The locomotion data has shown that vertical swimming does require faster locomotor speeds than other forms of locomotion; however, because of the small sample size of one turtle in deeper water, the truth of the hypothesis has not been proven one way or another. As with many of the hypotheses elaborated on below, although still not proven, this hypothesis is one that can be tested empirically, and should be tested by examining more extensive, more complete, future data sets, and should include data regarding important factors like water temperature and oxygen levels.
Pausing Behavior & Limb Frequency Cycles
The first stage of locomotion analysis in my snapping turtle research was a preliminary analysis of when the turtles paused, and when the turtles were locomoting. This analysis did not include when the turtle was breathing, and did not differentiate between different forms of locomotion. This analysis provided a framework for knowing when the turtles were pausing. The analysis was thus a preparatory step to simply get a sense for the overall behavior trends, before beginning the more specific and quantified analysis.
The major start to analyzing locomotion data came in recording limb frequency cycles for the three turtles. During continuous bouts of locomotion, the number of limb movements on one side (chosen to be the right side, which happens to be more visible in the footage) of the turtle (a logical proxy for the number of limb cycles) is recorded, as is the time duration of the bout. By dividing the movements into duration, a measure of cycles per second for limb cycle frequency is created. This is currently complete for Jawless and Snippy, and will not be performed for Lafayette.
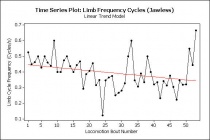
Plotting the LCF variable over time (using locomotion bout number as a chronological proxy for time), there is clearly a negative trend in Jawless's limb cycle frequency (See Fig. 3). This suggests an escape response, with high initial locomotion rates that decline as the turtle "relaxes," or in more scientific (and accurate) terms, decreases its overall stress level in response to a lack of human intervention and adjustment to the context. The Snippy data does not suggest an escape response, as Snippy's behavior is relatively constant throughout the video. Lafayette's footage would have been a useful means to establish a possible middle ground between Jawless and Snippy's data, but unfortunately, Lafayette sabotaged the analysis by sociopathically keeping her limbs out of the camera's view. Note that this hostility towards scientific advancement would not usually have been a major obstacle to making useful conclusions - however, given that even with Lafayette's data there would only be three turtles total to try to take down timelines for, the inter-turtle reliability of this analysis would be as slim as a Trionychid on a diet (in other words, nearly zero).
Even without Lafayette's support in our attempts to make inter-turtle analyses, there are still some important conclusions that can be drawn from what data we have. Firstly, Jawless demonstrated an escape response enacted by slowly reducing LCF over time - and providing some conclusions about how quickly the turtle can acclimate after disturbance. Secondly, pausing behavior plays a major role in the turtle's daily activity, which not only indicates that snapping turtles are incredibly lazy creatures, but suggests some important previously unknown behavioral mechanism that may also involve fish (see "Behavioral Novelties," below).
Methods of Locomotion
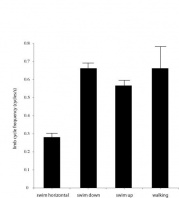
For these turtles, there are different methods of locomotion that can be differentiated into four behaviors: moving horizontally, swimming up, swimming down, and walking. To monitor limb frequency cycles differing between these behaviors, every bout of locomotion was recorded with the kind of behavior as well as limb cycle frequency.
From an ANOVA it appears swimming horizontally for Snippy was significantly slower than swimming vertically (in either direction) and walking (Fig. 5; this difference was the only significant one). However, because Jawless is in shallower water most of his behaviors can only be classified as horizontal swimming, as he has less of an "opportunity" to engage in vertical swimming.
Moreover, Jawless and Snippy both demonstrate little *visible* walking, but whereas Jawless is likely not engaging at all in this behavior, Snippy is likely walking with his limbs not visible, making it mostly impossible to document this behavior. Consequently, this allows the analysis to be possible for Snippy, but as most of Jawless' behavior is horizontal locomotion, an ANOVA-like test would not be possible or particularly relevant.
For Lafayette, the footage unfortunately turned out to be too murky for any significant analyses to be performed; moreover, for the majority of the footage, the limbs were not visible anyway. This may have been partly due to the fact that Lafayette was mainly walking, though there is no way of knowing for certain. While the behaviors were often apparent (it's hard to confuse vertical dives down with horizontal swimming), limb cycles could not be measured, which made the analysis futile.
A jawless turtle?!
There are many unusual features of behavioral and morphological traits demonstrated by the turtles. The most striking example comes from Jawless, the 39-pound snapper without a lower jaw. Certainly a well fed turtle! But how is this possible without a lower jaw? Do typical suction feeding mechanisms still work without half the feeding mechanism? We really can't answer this question, but it's certainly an interesting one to think about. Still, from what we have, we know there's something other than snapping that these turtles can do, and that it might be wise to listen to Chuck Smith's suggestion that we rename these "sucking turtles (Chelydra slurpentina)."
Unquantified Behaviors
The second key novelty demonstrated by the turtles was "the hitchhiking sunfish." During part of the footage collected from Snippy's Crittercam, a sunfish is visible, swimming along the turtle's neck as the turtle continues to swim, possibly unaware of the fish. While symbiosis between sunfish and snapping turtles would seem to be as probable as symbiosis between former President Clinton and a quarter-pounder Big-Mac, the sunfish certainly isn't phased by the turtle, and at times appears to be eating dead skin off the turtle's neck. Is this an example of some sort of symbiosis or commensalism? Unlike for Jawless' morphology, there is in fact a way to further examine the relationship between the sunfish and the turtle: more Crittercam! As the project is expected to get longer footage from more turtles during Summer 2009, future research will be able to examine this for evidence of a mutualism.
The third key behavioral/morphological novelty from the footage was fish activity surrounding the turtle. One could hypothesize that the one sunfish was just a particularly foolish (though clearly not appetizing enough for the turtle to notice) fish, and that most fish would be more wary of the turtles. The data suggests otherwise: all three turtles' footage includes resting periods during which large schools of fish aggregate in the vegetation surrounding the turtle and take no notice of the large carnivorous powerful-jawed predator watching them from its resting spot. Former research suggests that while juvenile snappers are active predators, adult snappers are in fact sit-and-wait predators, and while the behavior of these turtles in waiting for large schools of fish to slowly aggregate seems similar to such a feeding behavior, there is no predation documented in the footage. Why? Even more puzzling is that at one point, Snippy enters what appears to be a large fish "nursery", yet the fish take no notice, while the turtle swims carefully to avoid any collisions - a wonderful source for a snack, turned down - why? Are the turtles simply not hungry? Is there some sort of hope for a skin-cleaning behavior like that the sunfish may have exemplified? As with the sunfish, more data will help answer this question.
Current Analyses
Right now, I've finished analyzing breathing rates for all three turtles, a simple analysis of diving vs. pausing, and the data on limb locomotion rates for Jawless and Snippy. Although an analysis for Lafayette was attempted, unfortunately, Lafayette's footage does not show her limbs as much as would be necessary to complete an analysis, and so the locomotion data is restricted to the two turtles for which the limbs are visible. Consequently, analysis of the actual data is no longer "in progress." We even have a publication coming out this month in Herpetological Review (see below) discussing the incredible jawlessness of our jawless turtle. There's certainly major ways that this project could be continued and expanded, but to do so, more data collection will be required.
Related Publications
Landberg, T., Carlson, C. J., Abernathy, K., Luginbuhl, C. Gemme, P. and Mergins, Craig (in press) Natural History Notes: CHELYDRA SERPENTINA SERPENTINA L. (Eastern Snapping Turtle). SURVIVAL AFTER INJURY. Herpetological Review Dec. 2009
References
- ↑ Davenport J, Munks SA, Oxford PJ. A comparison of the swimming of marine and freshwater turtles. Proceedings of the Royal Society of London.Series B, Biological Sciences 1984 Feb. 22;220(1221):447-75.
- ↑ Zani PA, Claussen DL. Voluntary and forced terrestrial locomotion in juvenile and adult painted turtles, chrysemys picta. Copeia 1994 May 16;1994(2):466-71.
- ↑ Jackson DC. Respiration and respiratory control in the green turtle, chelonia mydas. Copeia 1985 Aug. 5;1985(3):664-7.1
- ↑ Lutz PL, Bentley TB. Respiratory physiology of diving in the sea turtle. Copeia 1985 Aug. 5;1985(3):671-9.
- ↑ West NH, Smits AW, Burggren WW. Factors terminating nonventilatory periods in the turtle, chelydra serpentina. Respiration Physiology 1989 9;77(3):337-49.
- ↑ Aerial and Aquatic Respiration in the Snapping Turtle, Chelydra serpentina http://www.jstor.org/stable/1565650