Roberta Engel
Post-Doctoral Research Associate
Severson Lab, University of Notre Dame, Feb. 2012 - present
Jockusch Lab, University of Connecticut Ph.D., Dec. 2011
E-mail:
rengel@nd.edu
Mailing address:
Dept.of Biological Sciences & Eck Institute for Global Health
Galvin Life Sciences Center
University of Notre Dame
Notre Dame, IN, U.S.A.
Contents
- 1 "Research is formalized curiosity. It is poking and prying with a purpose." (Zora Neale Hurston) and I would like to add "... that requires perseverance."
- 2 Diversification is a central theme in evolutionary biology...
- 3 Mosquito Genomics
- 4 Pseudoscorpions
- 5 Molecular Systematics: Sternophoridae
- 6 Molecular Systematics: Synsphyronus
- 7 Ultrastructures of the Chelicera
- 8 Pseudoscorpion Molecular Toolkit
- 9 Publications
- 10 Pseudoscorpion Projects
- 11 Scientific Awards & Fellowships
- 12 Science for All
- 13 Donate Science Books
- 14 Journal Club Coordinator, 2013 - present
- 15 Course Instructor
- 16 Lab Instructor
- 17 Teaching Collection Assistant Curator
- 18 Teaching Assistant
- 19 Connecticut Entomological Society
"Research is formalized curiosity. It is poking and prying with a purpose." (Zora Neale Hurston) and I would like to add "... that requires perseverance."
Diversification is a central theme in evolutionary biology...
and it is the diversification of arthropods that I find most fascinating. Divergence occurs at many levels; my research focus ranges from the individual to the genus level. I use genetics and genomics to answer basic questions about how diversity arises.
Mosquito Genomics
Currently, I am a Postdoctoral Research Associate in the Department of Biological Sciences and the Eck Instititue for Global Health at the University of Notre Dame. The Severson Lab studies the genetics and genomics of mosquitoes that transmit human diseases. I study genetic mechanisms that underlie complex traits associated with bloodfeeding in Culex pipiens and Aedes aegypti.
West Nile Virus is just one disease transmitted by the Culex pipiens complex and Aedes aegypti is a vector of multiple neglected tropical diseases including Dengue and Chikungunya. As no vaccine exists for these diseases, transmission is prevented through mosquito control. A more complete understanding of the genes and genetic pathways that regulate female reproductive behaviors will inform development and implementation of vector control methods. Our ability to generate informative genetic markers (SNPs) and infer quantitative trait loci (QTL) has increased dramatically with the advent of massively parallel sequencing technology (e.g., Illumina HiSeq2000). In addition, reference genomes are available for Cx. quinquefasciatus and Ae. aegypti.
The Culex pipiens species complex is broadly distributed. Cx. pipiens is found across temperate zones of the world while Cx. quinquefasciatus is restricted to subtropical and tropical regions. These two members of the complex are divergent in behavioral and physiological traits despite gene flow. One physiological trait that distinguishes Cx. pipiens from its sister taxon is its ability to enter reproductive diapause. Reproductive diapause is a complex trait triggered by environmental cues. Females mate but they do not take a bloodmeal and ovarian follicle development is arrested.
Aedes aegypti is a cosmopolitan species found primarily in urban areas across the tropics and subtropics. In the canonical life cycle of Ae. aegypti, the female mates, takes a required bloodmeal, and oviposits. Autogeny is the ability for females to oviposit without procuring a bloodmeal. This complex life history trait is facultative in Ae. aegypti and occurs at low incidence levels.
I am constructing a linkage map for both Cx. quinquefasciatus and Ae. aegypti using SNPs developed from Illumina HiSeq2000 datasets. The linkage maps will be used to infer QTL for reproductive diapause and autogeny. The high number of informative markers enables us to map the QTL regions at a fine scale. Candidate genes within QTL will be identified and genotype-phenotype associations will be tested through knockdown experiments.
Pseudoscorpions
Pseudoscorpions, also known as Chelonethida or False Scorpions, are an understudied order of arachnids. While not well known, Aristotle made note of pseudoscorpions as did the luminaries Linnaeus and Hooke (Micrographia 1665), and today they star on YouTube. Pseudoscorpions are found throughout the world in a wide range of microhabitats including leaf litter, animal nests, under rocks (even in the intertidal zone), beneath bark, and among the pages of books. One reason many are unfamiliar with pseudoscorpions is their size, most measuring less than 5 mm. This ancient lineage, the fossil record dates to the Middle Devonian (380MYA), currently comprises 25 families, 439 genera, and 3385 species. Pseudoscorpions of the World is a great online resource at http://museum.wa.gov.au/catalogues/pseudoscorpions.
Molecular Systematics: Sternophoridae
Sternophoridae is one of the smaller pseudoscorpion families. Presently, there are twenty described species which sort to three genera. Taxonomic work includes a revision of the family (Harvey 1985) and a survey the fauna of Florida (Hoff 1963). The three described species endemic to southeastern USA are the focus of my current work. Currently, populations of genus Idiogaryops endemic to southeastern USA are being surveyed. Molecular data will likely reveal cryptic diversity. Origins and evolutionary relationships among populations of Idiogaryops paludis, I. pumilus, and Garyops depressus will be inferred. In addition, the geographic ranges of these species provide an opportunity to test various phylogeographic hypotheses.
Molecular Systematics: Synsphyronus
Synsphyronus (Arachnida: Pseudoscorpiones: Garypidae) is a genus of pseudoscorpions found throughout Australia, New Zealand and New Caledonia (Harvey 1987). Currently, there are 24 described species but the diversity is much greater (unpublished data). This group has been collected beneath bark, in leaf litter, and under rocks in mesic and xeric regions. Specifically, I am interested in the origin and diversification of lineages that are endemic to the granite outcrops in southwestern Australia. Only one species, Synsphyronus elegans, has been described from the outcrops, and until recently it was known only from its type locality.
My research objectives included:
• to assess the diversity of Synsphyronus on the granite outcrops of southwestern Australia
• to describe species discovered on the outcrops
• to infer phylogenetic relationships among the rock-restricted lineages in southwestern Australia
• to estimate a species level phylogeny
I have been fortunate to have had three field seasons since starting my dissertation. During my 2004 and 2006 field seasons, I visited over 100 outcrops in southwestern Australia, sampling from the ~70 populations discovered. My sampling suggests that the diversity of Synsphyronus in southwestern Australia is greater than previously recorded. I traveled across Australia to localities recorded for twelve of the 22 Australian species of Synsphyronus during the Australian winter of 2008.
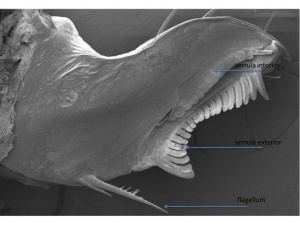
Ultrastructures of the Chelicera
Ultrastructures of the chelicera, including the serrula exterior, serrula interior, and flagellum blades, are taxonomically informative characters; I imaged these structures using scanning electron microscopy (SEM). The serrula exterior and interior teeth are heteromorphous. I have discovered papilla-like structures on the tips of the serrula exterior teeth. The serrula exterior is thought to be a grooming organ and the function of these ultrastructures warrants further exploration. Garypids typically have three spinose flagellum blades; the distal-most blade being the longest. The Synsphyronus samples I imaged had three blades (the flagellum shown is abnormal with only two blades). Many of the blades had structures that were more peg-like than spinose.
Pseudoscorpion Molecular Toolkit
Nuclear Genes
I have successfully isolated four nuclear genes (elongation factor 1- subunit α, wingless, actin 5C, and Internal Transcribed Spacer regions 1 and 2) from pseudoscorpions. I have designed new primers and modified published primers.
wingless, (wg)
The wingless primers used, wg1MP_F3 and wg1MP_R3, are modifications of those designed by Brower & DeSalle (1998) for use in beetles (pers. comm. M. Richmond).
wg1MP_F3 5’ GAR TGY AAR TGY CAY GGC ATG TCS GG 3’
wg1MP_R3 5’ ACY ICG CAR CAC CAR TGG AAI GTG CA 3’
Internal Transcribed Spacer regions 1 and 2 (ITS1, ITS2)
The ITS universal primers I used are found in Ji et al. (2003). I modified CAS28sB1 for use in pseudoscorpions.
CAS18sF1 5’ TAC ACA CCG CCC GTC GCT ACT A 3’
CAS28sB1mod 5’ TTT CCT CCG CTT ATT TAT ATG CTT AA 3’
Mitochondrial Genes
I recommend you visit Susan Masta's webpage Arachnid Mitochondrial Primers at http://web.pdx.edu/~smasta/primer_pages/Primer_front.html.
Two other sources for mitochondrial primers are the classic paper Evolution, weighting, and phylogenetic utility of mitochondrial gene sequences and a compilation of conserved polymerase chain reaction primers (Simon et al. 1994) and the more recent publication Incorporating molecular evolution into phylogenetic analysis, and a new compilation of conserved polymerase chain reaction primers for animal mitochondrial DNA (Simon et al. 2006).
Publications
Engel, R.S. (2012) Novel discovery of lamellar papillae on the grooming organ in Synsphyronus (Garypidae: Pseudoscorpiones). Arthropod Structure and Development. 41, 265-269.
in prep Engel, R.S., Backes, T., Musto, A., Mori, A. & Severson, D.W. Reproductive behavior including polyandry varies by strain in the yellow fever mosquito, Aedes aegypti.
in prep Engel, R.S. & Jockusch, E.L.. The evolution of EF1α, a multi-copy nuclear gene, in pseudoscorpions. target journal: Journal of Arachnology
Pseudoscorpion Projects
While at the UConn, I collected and imaged local fauna with the help of Evelize Codero (‘09), Elizabeth Riggles ('12) and Ashley Bonet ('12). Evelize imaged specimens collected by Dr. Raymond Pupedis, Senior Collections Manager, Division of Entomology, Yale Peabody Museum of Natural History. Chthonius and Microbisium are two genera found in Connecticut.The image of Chthonius was featured on UConn's 'Year of Science 2009' home page in the "What is it?" column.
Scientific Awards & Fellowships
2012 NSF Estimating Species Tree Workshop Travel Award
2010 EEB/CSMNH Award, James A. Slater Endowment
2008 NSF Doctoral Dissertation Improvement Grant
2008 Best Student Talk, Northeast Biological Graduate Students Conference
2007 Northeast Alliance at the University of Connecticut Summer Research Program Mentor
2006 Society of Systematic Biologists Mini-PEET Award
2006 SICB Fellowship for Graduate Student Travel
2006 EEB/CSMNH Award, James A. Slater Endowment
2005 Center for Conservation Biology, University of Connecticut (UConn)
2005 EEB/CT State Museum of Natural History Award (CSMNH), Lawrence R. Penner and James A. Slater Endowments
2005 NSF Graduate Research Fellowship, Honorable Mention
2005 American Arachnology Society
2004 Ecology & Evolutionary Biology Dept., UConn (EEB) Award, George Lamson Endowment
2004 NSF East Asia and Pacific Summer Institute Fellowship
2004 Australian Academy of Science Summer Institute Fellowship
Science for All
Recommended Reading
Darwin's Gift to science and religion Francisco J. Ayala
Parasite Rex Carl Zimmer
The Fever, how malaria has ruled humankind for 500,000 years Sonia Shah
This is Biology; the science of the living world E. Mayr
For Love of Insects T. Eisner
Broadsides from the Other Orders: A Book of Bugs S. Hubbell
The Diversity of Life E. O. Wilson
Reading the Forested Landscape: A Natural History of New England T. Wessels
A Sand County Almanac; and sketches here and there A. Leopold
The Immortal LIfe of Henrietta Lacks R. Skloot
The Botany of Desire M. Pollan
Why Evolution Is True J. Coyne
Children's Literature
As an evolutionary biologist and a former elementary school teacher, I am interested in the literature that is available to school age children. Titles in my collection include,
Jackson, Ellen The Tree of Life, The Wonders of Evolution
Lasky, Kathryn One Beetle Too Many, The Extraordinary Adventures of Charles Darwin
Markle, Sandra Animals Charles Darwin Saw, An Around-the-World Adventure
Manning & Granstrom What Mr. Darwin Saw
Peters, Lisa W. Our Family Tree, An Evolution Story
Schanzer, Rosalyn What Darwin Saw, The Journey that Changed the World
Sis, Peter The Tree of Life
Donate Science Books
Pay your books forward. Consider donating books to groups such as Books Through Bars [www.booksthroughbars.org/]. I have accumulated several used biology textbooks and found a local group that will put them to use!
Journal Club Coordinator, 2013 - present
Diapause Journal Club
Recently (Fall 2013), I realized several investigators in the Biological Science Department at the University of Notre Dame are studying diapause in non-model arthropod systems and decided to form an ad hoc discussion group. We meet once a month to discuss recent publications, present preliminary data, and share research methods. Currently, all in the group work with arthropods but anyone is welcome to join the conversation.
Apr. 2, 2014
Diapause in leps, cont. Sean will share a model he is developing.
Feb. 19, 2014
Diapause in leps
Jan. 22, 2014
Discussion will grow out of a recent publication that focuses on the role of sRNAs in diapause pathways (Reynolds et al. 2013). Susanta has agreed to join us and will provide some background on his sRNA studies. (see Behura 2007 for a review of microRNAs).
Dec. 11, 2013
Paul will present gene expression data from his work on the induction of diapause in Culex pipiens.
Nov. 18, 2013
Our meeting will focus on Microarrays Reveal Early Transcriptional Events during the Termination of Larval Diapause in Natural Populations of the Mosquito, Wyeomyia smithii. (Emerson et al. 2010).
Nov. 6, 2013
Greg will talk about his work with Rhagoletis. Recommended reading: Developmental trajectories of gene expression reveal candidates for diapause termination: a key life-history transition in the apple maggot fly Rhagoletis pomonella. (Ragland et al. 2011).
Course Instructor
Field Entomology
Lab Instructor
General Entomology
Medical Entomology
Teaching Collection Assistant Curator
Entomology Teaching Collection Assistant
Teaching Assistant
Evolutionary Biology
General Ecology
Principles of Biology I & II (majors)
Foundations of Biology (non-majors)
Connecticut Entomological Society
http://www.insectsingers.com/ces/ces.html
2009-2010 President
2008-2009 Vice President
2007-2008 Vice President
2009-2010 Speaker Series
May: K. Zyko (CTDEP), L. Saucier (CTDEP), M. Thomas (CAES) & C. Maier (CAES); The Natural Diversity Database (DEP) and Status Update on Connecticut State-listed Insects
April: Kathy Hill & David Marshall, UConn; A Cicada Travelogue
March: Jan Conn, Wadsworth Center; Recent divergence or deep population structure? Complexities of the primary neotropical malaria vector Anopheles darlingi (Diptera: Culicidae)
February:Jadranka Rota, Smithsonian Institution; Systematics and defensive behaviors of metalmark moths (Lepidoptera: Choreutidae)
January: Kentwood Wells, Dept. Chair, EEB, UConn; Beetles, Bees, and Butterflies: Charles Darwin and the Study of Insects
November: Patrick Getty, UConn; Fossil Trackways of Modern Basal Insects
October: Andrew Stoehr, Yale; Variation in Melanism in Pierid Butterflies
September: Kirby Stafford III, CAES; Asian Longhorned Beetle: A Threat to our Trees